Neurodegenerative Diseases
Alzheimer’s Disease
FAST FACTS
Over 6.2 million Americans, age 65 and older are living with Alzheimer's dementia today
APOE4 is the strongest and most reproducible genetic risk factor for late-onset Alzheimer’s disease (LOAD)
Of the people diagnosed with Alzheimer’s:
%
have 1 copy of the APOE-e4 gene
%
have 2 copies of the APOE-e4 gene
AD patients with APOE4:
Accelerated onset of dementia
Worse memory performance
Overview
Late Onset AD (LOAD)
Late onset AD (LOAD) is a complex disease and the variability in the genetic risk and molecular profiles amongst AD patients presents significant challenges for drug development. Advancement in LOAD therapy will require the development and validation of new drug targets, including those tailored to sub-group/s of patients with specific risk factors. As such, the focus has shifted to new potential culprits, including gene-targets of LOAD. Consistently, APOE has emerged as a target for LOAD treatment.
APOE as a target
A major risk determinant of LOAD is the apolipoprotein E (APOE) gene, There are three primary allelic variants, e2, e3, and e4. APOEe2 is associated with lower disease risk, while the APOEe4 allele is the strongest and most reproducible known genetic risk factors for LOAD. Carrying one APOEe4 allele increases the risk of LOAD 3-4-fold and carrying two increases risk 9-15-fold. Further, AD individuals carrying APOEe4 have an earlier onset of dementia and worse memory performance.
Lifetime risk of Alzheimer’s Disease based on number of APOE4 copies
- Zero: ~9% (ApoE3/3, ApoE2/3 Genotype)
- One: ~30% (ApoE3/4, ApoE2/4 Genotype)
- Two: ~50-90% (ApoE4/4 Genotype)

Given the role APOEe4 plays in numerous AD-related pathways (tau, amyloid plaques, neuroinflammation), APOEe4 has emerged as a promising AD drug target
ApoE4
APOE4 impacts key drivers of AD pathogenesis
Increase Abeta aggregation
Decrease lipid/cholesterol transport and clearance
Decrease cellular function
Decrease Abeta clearance
Increase inflammatory response
Decrease Insulin and VEGF signaling
Increase tau phosphorylation and tau tangle formation
Decrease glucose and mitochondrial metabolism
Decrease synaptic and neuronal function
CLRI-002
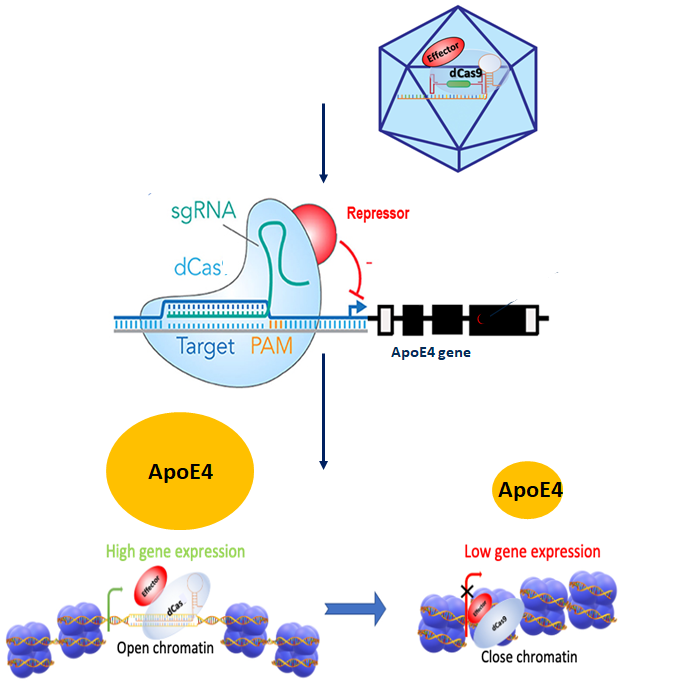
CLRI-002 is designed to lower the expression of APOE4 without the side effects of turning off APOE4 completely. The resulting impact is a reduction of amyloid and tau pathology and a decrease in neuroinflammation
While several groups are developing drugs to eliminate APOEe4 (via knockdown or clearance), other small studies demonstrate that complete elimination of APOEe4 may be detrimental, supporting the evolutionary maintenance of this gene. Therefore, a modern and precise LOAD therapeutic should take into consideration carrier status and strive to lower APOE4 to prevent the downstream pathogenic effects.
CLAIRIgene has developed epigenome editing tools to downregulate APOEe4 expression precisely and in allele-specific manner. Using the developed platform highlighted below, we were able to effectively and specifically modulate APOE expression by targeted modification of the epigenome landscape within APOE locus. Our gene-delivery system is based on the optimized and improved, proprietary Adeno-Associated Viral (AAV) vector. Our epigenome therapy strategy for fine-tuning of APOE expression based on dCas9 technology is translational toward the development of a therapeutics approach to prevent and/or delay LOAD onset.
Citation: https://www.biorxiv.org/content/10.1101/2023.04.14.536951v1
Publication is currently under revision in Nature Communication
Parkinson’s Disease
FAST FACTS
Nearly 90,000 people in the U.S. are diagnosed with Parkinson’s Disease
Nearly one million people in the U.S. are living with Parkinson's disease (PD)
%
percent of Parkinson's cases to a direct genetic cause
Parkinson’s disease (PD) is the second most common neurodegenerative disorder in the world, nevertheless, currently there is no effective treatment to prevent PD or to halt its progression. The SNCA gene has been implicated as a highly significant genetic risk factor for PD. In addition, accumulating evidence has suggested that elevated levels of wild-type a-synuclein are causative in the pathogenesis of PD. To date, a-synuclein, encoded by the SNCA gene, is one of the most validated and promising therapeutic targets for PD. Moreover, manipulations of SNCA levels have demonstrated a beneficial impact. However, neurotoxicity associated with robust reduction of SNCA levels was reported in studies that utilized RNAi tools to target directly SNCA transcripts. To better control levels of SNCA, our founders, Drs. Chiba-Falek and Kantor created a system to tightly regulate the expression of SNCA transcription.
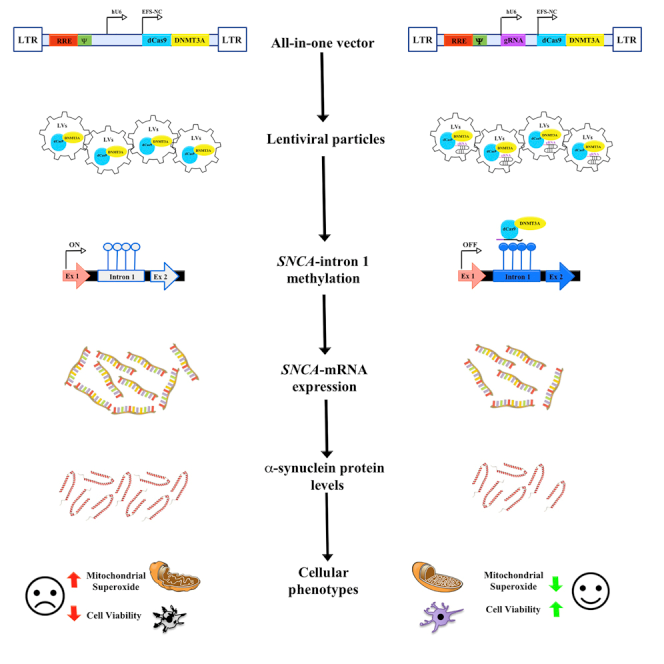
CLAIRIgene’s Approach
Elevated levels of SNCA have been implicated in the pathogenesis of Parkinson’s disease (PD), while normal physiological levels of SNCA are needed to maintain neuronal function. We developed new therapeutic strategies targeting the regulation of SNCA expression. To achieve that, we targeted DNA methylation at SNCA intron 1. DNA methylation at SNCA intron 1 is an attractive target for fine-tuned downregulation of SNCA levels, as it regulates SNCA transcription, and PD brains showed differential methylation levels compared to controls. To target DNA methylation in intron 1 of SNCA, we developed a system, comprising an all-in-one lentiviral vector, carried CRISPR/dCas9- fused with the catalytic domain of DNA-methyltransferase 3A (DNMT3A). Applying the system to human induced pluripotent stem cell (hiPSC)-derived dopaminergic neurons from a PD patient with the SNCA triplication resulted in fine downregulation of SNCA mRNA and protein mediated by targeted DNA methylation at intron 1. Furthermore, the reduction in SNCA levels by the guide RNA (gRNA)-dCas9-DMNT3A system rescued disease-related cellular phenotype characteristics of the SNCA triplication hiPSC-derived dopaminergic neurons, e.g., mitochondrial ROS production and cellular viability. Thus first-generation system using LV (licensed to Seelos Therapeutics) has been most recently evolved into second and third generation platform utilized AAV vector as the delivery vehicle.
In our new program (CLRI-005) we demonstrated the high potential of this target sequence combined with the AAV-CRISPR/dCas9 technology as a novel epigenetic-based therapeutic approach for PD. This is available for licensing
Kantor B, et al. (2018) Downregulation of SNCA Expression by Targeted Editing of DNA Methylation: A Potential Strategy for Precision Therapy in PD. Molecular Therapy. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6224806/
Tagliafierro L, Ilich E, Moncalvo M, Gu J, Sriskanda A, Grenier C, Murphy SK, Chiba-Falek O, Kantor B. Lentiviral Vector Platform for the Efficient Delivery of Epigenome-editing Tools into Human Induced Pluripotent Stem Cell-derived Disease Models. J Vis Exp. 2019 Mar 29;(145):10.3791/59241. doi: 10.3791/59241. PMID: 30985756; PMCID: PMC7144461. https://pubmed.ncbi.nlm.nih.gov/30985756/
Tagliafierro L, Chiba-Falek O. Up-regulation of SNCA gene expression: implications to synucleinopathies. Neurogenetics. 2016 Jul;17(3):145-57. doi: 10.1007/s10048-016-0478-0. Epub 2016 Mar 7. PMID: 26948950; PMCID: PMC4907864. https://pubmed.ncbi.nlm.nih.gov/26948950/
Lewy Body Dementia
FAST FACTS
Dementia with Lewy bodies is estimated to affect 1.4 million people in the United States.
Mutations in genes known as SNCA can cause dementia with Lewy bodies
%
It accounts for about 5 percent of all dementia cases in older individuals and is the second most common dementia after Alzheimer disease.
Lewy body dementia (LBD) is a progressive brain disorder that affects thinking, movement, behavior, and mood. It is caused by the buildup of abnormal protein deposits called Lewy bodies in the brain.
LBD is not the same as Parkinson’s, but the two are closely related: LBD causes some or all of the motor symptoms of Parkinson’s. More than 1 million people in the U.S. are affected by Lewy body dementia, according to the Lewy Body Dementia Association. There are two main types of LBD: dementia with Lewy bodies (DLB) and Parkinson’s disease dementia (PDD). DLB and PDD are very similar, but the symptoms appear in a different order depending on which type of LBD a person has.
In DLB, dementia symptoms (such as memory loss, confusion, and difficulty thinking) usually appear first. Motor symptoms (such as tremors, stiffness, and slowness of movement) may develop later.
In PDD, motor symptoms usually appear first, followed by dementia symptoms.
Other symptoms of LBD can include:
- Visual hallucinations
- Sleep disturbances
- Falls
- Depression
- Anxiety
- Difficulty swallowing
- Constipation
- Urinary incontinence
There is no cure for LBD…we hope to change that
Patient and Caregiver Resources:
To learn more about Alzheimer’s Disease, visit
To learn more about Parkinson’s Disease, visit
To learn more about Lewy Body Dementia, visit https://www.lbda.org/