Platform
Problem:
Low efficacy of delivery system often requiring duel AAV vectors
Problem:
Inadequate and inefficient combination of bulky and multicomponent CRISPR/Cas system
Problem:
Inadequate and inefficient combination of CRISPR/Cas effector system pairing
CLAIRIgene:
Novel AAV-CRISPR/deactivated(d)Cas9-repressor platform created via optimizedCas-repressor pairing screening cascade
Overview
CLAIRIgene’s epigenome editing represents a novel appealing approach for gene therapy mediated by altering expression of a specific gene based on CRISPR/dCas9 technology. Genome editing approaches change the genetic code via DNA cleavage and are prone to introduce new mutations. However, epigenome editing uses deactivated nuclease, thus, the DNA remains intact and no changes to the DNA sequence. This technology allows us to modify epigenetic markers to control the expression of disease-associated genes. Our allelic discrimination approach, combined with the utility of dCas9- variant is innovative and allows more fine-tuned regulation of allele expression.
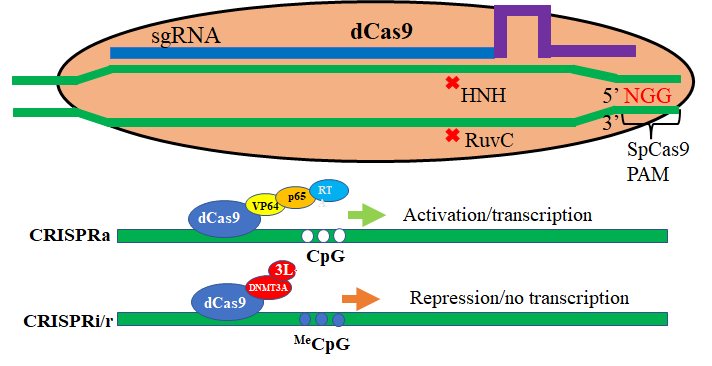
Activation and repression of a target gene: RNA-programmed gene activators can be assembled through the direct fusion of dCas9 with the transcriptional activators VP64, or indirect- via MS2 binding motif- with transcriptional activators p65-HSF, NF-kb and others (not shown here- the histone acetyltransferase enzyme p300 or the DNA demethylase Tet1). Alternately, transcriptional repressors such as KRAB, de-novo methyltransferase 3A and the histone demethylase LSD1 can be fused either directly or indirectly to repress a target gene.
Rittiner, J, Cumaran, M, Malhotra, S, and Kantor, B Therapeutic modulation of gene expression in the disease state: development of next-generation epigenetic drugs paired with viral vector delivery Front Bioeng Biotechnology. 2022 Oct 17;10:1035543. doi: 10.3389/fbioe.2022.1035543 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9620476/
Kantor B and Chiba-Falek O. Lentiviral vectors as the delivery vehicles for transduction into iPSCs: shortcomings and benefits 2021. Advances in Stem Cell Biology. Book Chapter (Methods in iPSC Technology Volume 9 in Advances in Stem Cell Biology 2021, Pages 79-100).
Dong, W and Kantor, B. Lentiviral Vectors for Delivery of Gene-Editing Systems Based on CRISPR/Cas: Current State and Perspectives. Viruses 2021, 13, 1288. https://doi.org/10.3390/v13071288
Rittiner J, Moncalvo M, Chiba-Falek O , Kantor B. Gene-editing technologies paired with viral vectors for advancing basic and translation research of Neurodegenerative Diseases. Front. Mol. Neurosci., 12 August 2020 https://www.frontiersin.org/articles/10.3389/fnmol.2020.00148/full
For efficient drug delivery packaging, CLAIRIgene has established and validated the Adeno-Associated Virus (AAV)-CRISPR/deactivated(d)Cas9-repressor system. AAV vectors are the leading platform for gene delivery; however, the capacity of the AAV is small, ~4.7kb genome size, which is a limitation to package the bulky multicomponent CRISPR/Cas system into a single AAV43. Co-delivering the CRISPR/Cas9 system with separate AAV vectors result in inefficient and inadequate delivery.
Our novel all-in-one vector technology circumvents several challenges related to gene therapy: (i) optimized AAV backbone for high packaging and delivery efficiencies of the CRISPR/Cas9 components; (ii) suitability for a broad range of cellular tropisms; (iii) low cytotoxicity, immunogenicity; (iv) long-term, sustainable expression of the vector to ensure the durability of the epigenetic changes.